Generalized epilepsy
generalized absence EPILEPSY
YOUR PRECLINICAL AND CLINICAL ROADMAPs. ALIGNED.
A translational model of absence seizures
Genetic absence epilepsy rat from Strasbourg (GAERS) is a selectively inbred strain of Wistar rats displaying spontaneous spike-and-wave discharges (SWD). The GAERS has become a reference model for absence epilepsy for more than 30 years, since these rats present behavioral, electrophysiological and pharmacological features of human absence seizures.
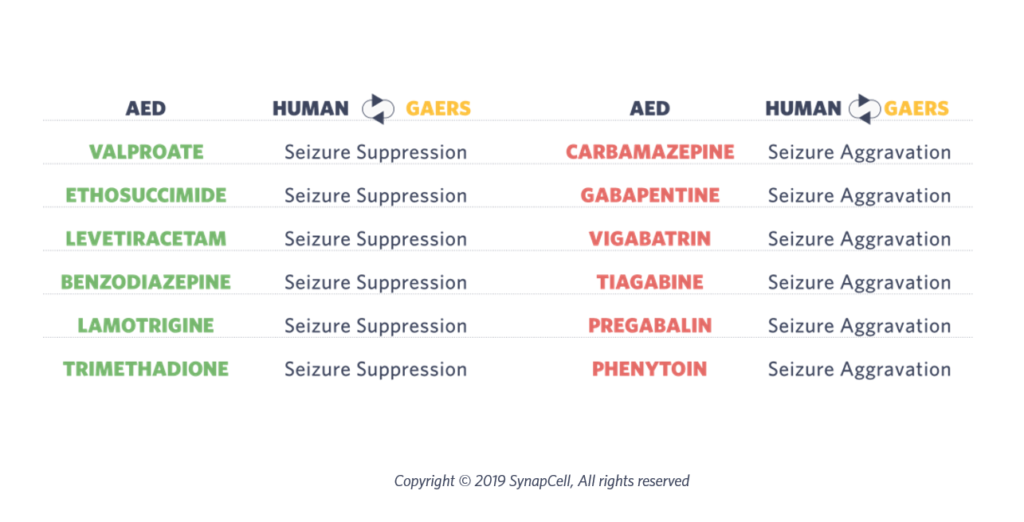
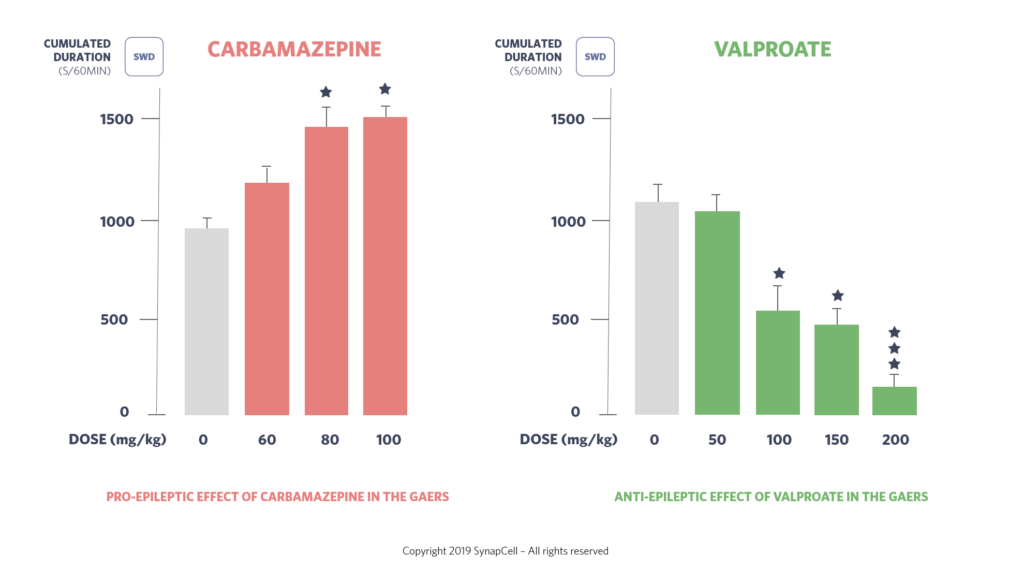
The pharmacology of the GAERS model is indeed just like the human’s. For instance, valproate, ethosuximide and levetiracetam are equally effective in both human patients and GAERS.

SYNAPCELL'S WORLD-EXCLUSIVE MODEL

- COMBINE
- DERISK
- SECURE
- DECIDE
the gaers pharmacology
translates into the clinic
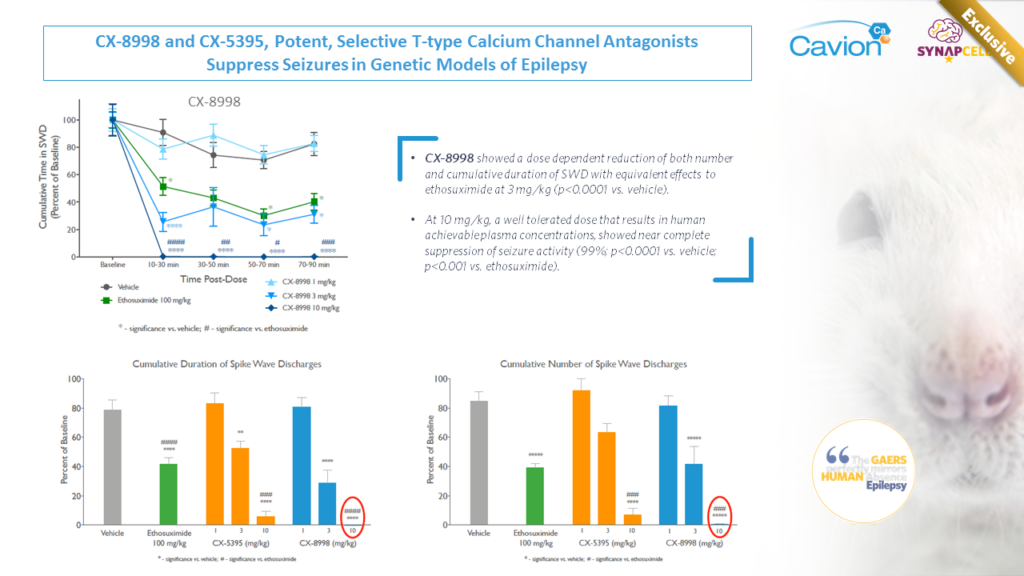
JOINT WORK WITH CAVION
Cavion’s T-Type Calcium Channel Modulator CX-8998 demonstrated Superior-effect to Current Standard of Care in Suppressing Absence Seizures in SynapCell’s Genetic Absence Epilepsy Model (GAERS)
JOINT PUBLICATION WITH PFIZER
PF-06372865 dose-dependently reduced the occurrence of SWD in the GAERS model.
PF-06372865 is a positive allosteric modulator (PAM) of α2/3/5 subunit-containing GABAA receptors with minimal activity at α1-containing receptors, which are believed to mediate many of the adverse events associated with benzodiazepines.
The aim of this study was to assess the antiepileptic effect of PF-06372865 in a preclinical model of absence seizures, the GAERS rat.
Let's talk about your next project

Bâtiment SYNERGY
ZAC ISIPARC
38330 Saint Ismier – France
OPEN INNOVATION
CAPABILITIES
SYNAPCELL 2022 © – All Rights Reserved